Thursday, August 29, 2013
Tuesday, August 27, 2013
A 20-something with palpitations and dizziness. What action will you take?
I have posted on this topic before, but whenever I think that readers completely understand it, I find there are still many who do not. So I think it is worth the repetition. Becoming facile at reading ECGs is a matter of repetition and practice.
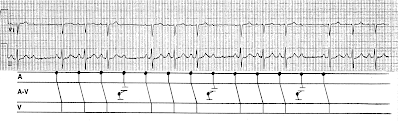
A 20-something calls 911 for palpitations and dizziness, arrives in the ED and has this ECG recorded. What are you going to do?
Below I have put arrows on the R-R interval that is shortest.
 |
| This very short R-R interval is approximately 160 ms! |
1. What are you going to do?
2. (Less important) What is the diagnosis?
3. What are you definitely not going to do?
Answers
1. Cardiovert. Cardioversion will treat it effectively, whatever the etiology. This is so fast that it needs rather urgent therapy. Any medication could be ineffective or, worse, dangerous. (It may be reasonable, but much less preferable, to treat with procainamide or another Type 1 antiarrhythmic if for some reason cardioversion cannot be done).
2. The rate is fast and the rhythm is irregularly irregular, so this is Atrial fibrillation. With the very fast ventricular response and the bizarre, polymorphic QRS, it is nearly pathognomonic for atrial fibrillation in the setting of WPW. The impulses conduct variably and very fast down the accessory pathway because it has a very short refractory period. Whenever the shortest R-R interval is less than 240 ms, there is a high risk of degeneration into ventricular tachycardia or fibrillation, especially if AV nodal blockers are given. In fact, the morphologies during this 1.6 seconds of extremely fast rate are so different that they may, in fact, be a short run of polymorphic ventricular tachycardia, though this is impossible to know.
3. You are not going to give any AV nodal blockers (no Digoxin, Adenosine, Calcium Channel Blocker, or even Beta Blocker)
Finally, this prohibition against AV nodal blockers only applies to Atrial Fibrillation with WPW. It does not apply to:
1. WPW with a regular tachycardia, even if wide (Patients with AV reciprocating tachycardia (antidromic reciprocating tachycardia) respond to AV nodal blockers. They are not dangerous and are very helpful
2. Atrial fibrillation with a monomorphic wide complex, and no very short R-R intervals, is not WPW. It is atrial fibrillation with aberrancy, usually RBBB or LBBB, and these may be safely slowed with AV nodal blockers such as Diltiazem or a Beta Blocker.
I do not have followup on this case.
For more on this topic and more interesting cases, see this post, and this post.
Monday, August 26, 2013
Pseudo Type II Second Degree AV Block Induced by Concealed Premature Junctional Complexes (PJCs) (contributed by K. Wang)
This patient is running on a treadmill:
The stress test was stopped for fear that it was induced by ischemia.
This tracing was also recorded during the stress test:
Here we have the first ECG (Figure 1) with a ladder diagram:
Thus, what appears to be a troublesome situation turned out to be simple manifestations of frequent concealed PJCs, which are benign
A similar diagram for PACs can be seen here at Medscape ECG of the Week posted on 3/7/2012:
 |
| Figure 1. There are frequent apparently non-conducted p-waves. The PR interval is not lengthening, so this is not AV Wenckebach phenomenon. It appears to be 2nd degree AV block, Mobitz Type II. |
The stress test was stopped for fear that it was induced by ischemia.
This tracing was also recorded during the stress test:
Here we have the first ECG (Figure 1) with a ladder diagram:
Thus, what appears to be a troublesome situation turned out to be simple manifestations of frequent concealed PJCs, which are benign
A similar diagram for PACs can be seen here at Medscape ECG of the Week posted on 3/7/2012:
Instructions for using subtleSTEMI iPhone app
Some have asked for instructions in using the app:
1.
The warning states that it should only be used
when the differential diagnosis is
normal variant anterior ST elevation (early repolarization) vs. subtle anterior
STEMI
2.
Questions (these were exclusions from the study):
a. Is
there bundle branch block (if so, then the formula does not apply, answer must
be “no”
b. Is
the T-wave inverted in any of V2-V6, but not due to “benign T-wave inversion”. (If yes, then think STEMI and do not use
formula)
i. In
general, if there is any T-wave inversion, the formula does not apply
ii. If
you are expert, then you can differentiate benign T-wave inversion from
ischemic, and you shouldn’t need the formula anyway
c. Is
the ST segment elevated > 5 mm in any lead?
i. Then
it should be considered to be STEMI (If
yes, then think STEMI and do not use formula)
d. The
question: "is terminal QRS distortion present (absence of both S-wave and
J-wave in V2 or V3)" should read V2 and V3
i. Early
repol does not have such terminal QRS distortion. Anterior STEMI commonly does. (If yes, then think STEMI and do not use
formula)
e. Do
any of leads V2-V6 have a convex ST segment ?
This means upward convexity.
i. Early
repol should almost always have upward concavity in these 5 leads. (If yes, then think STEMI and do not use
formula)
f. Significant
ST depression in leads II, III, aVF?
Early repol never has reciprocal ST depression
i. (If
yes, then think STEMI and do not use formula)
g. Is
there ST depression in V2-V6? Early repol
never has precordial ST depression
i. (If
yes, then think STEMI and do not use formula)
h. Is
there Q-waves in V2-V4? (this should say
“significant, pathologic Q-waves”. Early
repol never has pathologic Q-waves
i. (If
yes, then think STEMI and do not use formula)
If the answer to all these is no, the app takes you to the formula, where you enter:
1) ST elevation in millimeters, measured at 60 milliseconds after the J-point in lead V3
2) the computerized QTc measurement
3) the R-wave amplitude as measured in lead V4.
If the internal (black box) formula returns a value > 23.4, the app will say that it is STEMI. If it returns a value < 23.4, it will say "Early repolarization".
This is the formula: ([1.196 x ST segment elevation 60 ms after the J point in lead V3 in mm] +
[0.059 x QTc in ms] minus [0.326 x R-wave amplitude in lead V4 in mm])
This is the study from which it comes:
The accuracy, sensitivity for STEMI and specificity for STEMI are about 90%
Saturday, August 24, 2013
K. Wang Video: Aortic Dissection? STEMI? Both? (5 minute video)
This is a fine short summary of the topic.
One disclaimer: At the end of the talk, K. states that ST segment depression in lead I in inferior STEMI indicates right ventricular MI. I have data that would refute that claim.
One disclaimer: At the end of the talk, K. states that ST segment depression in lead I in inferior STEMI indicates right ventricular MI. I have data that would refute that claim.
Friday, August 23, 2013
Diffuse ST elevation and Chest pain in an Agitated Middle-aged male: Anterior STEMI or Takotsubo Stress Cardiomyopathy?
A male in his 40's was discharged from the ED after evaluation for chronic back pain. He was denied opiate medication and was very upset with this. Shortly after discharge, he developed acute chest pain, diaphoresis and pre-syncope. EMS was called and they recorded a BP of 50. They recorded this ECG:
The medics activated the cath lab prehospital.
He arrived in the ED tearful and agitated and required much consoling. His coronary risk factors were smoking and hypertension. This ECG was recorded at time zero:
 |
| No significant change |
Here is his previous ECG:
A bedside echocardiogram showed poor function, but was not more specific. The patient took prolonged persuasion to consent to going to the cath lab.
This ECG was recorded at time 30 minutes:
 |
| The hyperacute T-waves are resolving, as is the ST elevation. The ischemia is greatly improving. |
The patient was taken to the Cath Lab and found to have no significant coronary disease. Troponin I peaked at 0.3 ng/ml. A formal echocardiogram showed severely decreased LV systolic function with the estimated left ventricular ejection fraction at 20-25 %. There were wall motion abnormalities in mid to distal LV wall segments, with sparing of the basal LV segments, all highly suggestive of Takotsubo type stress induced cardiomyopathy.
How to suspect takotsubo stress cardiomyopathy from the presenting symptoms, signs and ECG
First, it is believed to by caused by diffuse small vessel ischemia due to catecholamines, and thus has the same electrophysiologic substrate as STEMI. It affects the entire heart except the base, resulting in diffuse circumferential wall motion abnormalities that only spare the base (top) of the heart. These diffuse wall motion abnormalities (lateral, posterior, inferior, septal, anterior, apical) result in systolic "apical ballooning" which looks like a Japanese octopus trap (a takotsubo).
Of course, the clinical presentation can help to suspect this: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3214344/
It is most common in postmenopausal women and is associated with severe emotional upset (stress) or severe physiologic stressors such as respirtory failure.
Stress Cardiomyopathy (Apical Ballooning syndrome, or Takotsubo cardiomyopathy) only presents with ST elevation in about 1/3 of cases, but when it does, it is one of the most difficult mimics of anterior STEMI, and often the only way to tell the difference is to do an angiogram.
ECG Differentiation
A review and analysis of the literature in ECG differentiation of the two entities (1-6) reveals that an analysis of ST elevation vector is a good predictor of anterior STEMI vs. stress cardiomyopathy. In anterior STEMI, whether proximal, mid, or distal LAD occlusion, there is ST elevation in V1 (1 mm measured at 80 ms after the J-point) in about 80% of cases, whereas this is present in 20% of cases of SCM. SCM also has a negative ST segment in aVR and is more likely to have ST elevation in inferior leads, or at least absence of ST depression in inferior leads (however, 40% of anterior STEMI lack inferior ST depression). Finally, precordial ST elevation in SCM is more pronounced in V3-V5 vs. V2-V4.
Putting these all together, it is apparent that the ST vector in anterior STEMI is more commonly anterior and superior (V1-V4), without STE in inferior leads, whereas in SCM the ST Vector it is more inferior and lateral (V2-V5, with STE in inferior leads and ST depression in aVR). This correlates with the location of wall motion abnormalities in SCM (diffuse and toward the apex, similar to pericarditis, including inferior and lateral walls) vs. anterior STEMI (anterior as well as septal-apical).
This Case
1. The patient was very stressed and emotionally upset and had just been denied opiates for his pain
2. There is diffuse ST elevation: anterior, inferior and lateral. Of course this could be a proximal LAD occlusion with a wraparound LAD such that all 3 walls are supplied by the LAD.
3. Absence of ST elevation in lead V1
4. ST depression in aVR
Final Assessment:
One might do a formal echo to look for apical ballooning, and this may help in diagnosing Takostubo, but given the present state of the art, I do not believe that there was enough information to confidently exclude anterior STEMI (LAD occlusion) based on clinical, ECG, and echo characteristics without doing an angiogram.
1.
Tamura
A et al. A New Electrocardiographic
Criterion to Differentiate Between Takotsubo Cardiomyopathy and Anterior Wall
ST-Segment Elevation Acute Myocardial Infarction. Am J Cardiol Sept 2011; 108(5):630-633.
2. Ogura
R, et al. Specific findings of the standard 12-lead ECG in patients with “takotsubo” cardiomyopathy: comparison with the findings of acute anterior myocardial infarction. Circ J 2003;67:687–
690.
3.
Inoue
M, et al.
Differentiation between patients with takotsubo cardiomyopathy and those with
anterior acute myocardial infarction. Circ J 2005;69:89 –94.
4. Bybee KA, et al. Electrocardiography cannot reliably differentiate transient left
ventricular
apical ballooning syndrome from anterior ST-segment elevation myocardial infarction. J Electrocardiol 2007;40:38.e1–38.e6.
5. Jim MH, et al. A new ECG criterion to identify takotsubo cardiomyopathy from
anterior
myocardial infarction: role of inferior leads. Heart Vessels 2009;24:124–130.
6. Kosuge M, et al. Simple and accurate electrocardiographic criteria to differentiate
takotsubo
cardiomyopathy from anterior acute myocardial infarction. J Am Coll Cardiol 2010;55:2514 –2516.
Subscribe to:
Posts (Atom)